Abstract
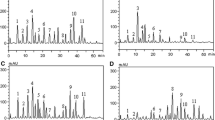
The main objective of this current study was to assess the protective role of Cyperus rotundus L. (CR) extract against oxidative stress, neurotoxicity, and inflammation induced by esfenvalerate in rats. The total phenol (TP) and total tannins (TT) were estimated by Folin ciocalteu and total flavonoids were evaluated by aluminum chloride methods. The methanol: acetone: H2O with ratio 2:2:1 extract of C. rotundus tubers was determined antioxidant activity by DPPH, ABTS•+scavenging activities, and ferrous chelating, reducing power activities assays. Antioxidant activities of C. rotundus tuber extract exhibited 224.25, 191.47, and 218.77 μg/ml against 2,2-diphenyl-1-picrylhydrazyl (DPPH•), and 2,2-azinobis (3-ethylbenzothiazoline-6-sulfonic acid) (ABTS•) radicals and Fe2+-chelating, respectively expressed as IC50 while reducing power showed 119.88 μg/ml expressed as EC50. C. rotundus tuber extract’ analysis showed a presence of several phenolic and flavonoid compounds identified by HPLC. Albino Wistar rats were divided into normal control, C. rotundus alone treated esfenvalerate, and treated (Esfenvalerate + CR) groups. The dose of C. rotundus extract was100 mg /kg BW, while the dose of esfenvalerate was 0.533 mg/kg BW orally. Administration of esfenvalerate decreased the levels of brain reduced glutathione (GSH), and paraoxnase-1(PON-1), and decreased acetylcholinesterase activity along with increasing the levels of brain malondialdehyde (MDA) and nitric oxide (NO), furthermore, increased serum tumor necrosis factor-alpha (TNF-α), adiponectin, and lipocalin-2. On the other hand, treatment with C. rotundus extract significantly showed a protective effect against esfenvalerate by ameliorating levels of antioxidant enzymes, acetylcholine esterase, and inflammatory markers. The present study elicited a prophylactic effect of C. rotundus against neural damage induced by esfenvalerate in experimental rats.
Similar content being viewed by others
References
Al-Omar MA, Beedham C, Alsarra IA (2004) Pathological roles of reactive oxygen species and their defence mechanisms. Saudi Pharm J 12(1):01–18
Arnao MB, Cano A, Acosta M (2001) The hydrophilic and lipophilic contribution to total antioxidant activity. Food Chem 73(2):239–244
Baconi DL, Bârca M, Manda G, Ciobanu AM, Balalau C (2013) Investigation of the toxicity of some organophosphorus pesticides in a repeated dose study in rats. Romanian J Morphol Embryol 54(2):349–3456
Bahi A, Necib Y (2014) Hepatoprotective and antioxidant activity of aqueous extract of Cyperus rotundus rhizome against mercuric chloride induced oxidative stress in rats. Int J Pharm Sci Rev Res 27(1):117–123
Bashir A, Sultana B, Akhtar FH, Munir A, Amjad M, Hassan Q (2012) Investigation on the antioxidant activity of Dheela Grass (Cyperus rotundus). African J Basic Appl Sci 4(1):1–6
Belewu MA, Abodunrin OA (2006) Preparation of Kunnu from unexploited rich food source: tiger nut (Cyperus rotundus). World J Dairy Food Sci 1(1):19–21
Belewu MA, Belewu KY (2007) Comparative physico-chemical evaluation of tiger-nut, soybean and coconut milk sources. Int J Agric Biol 9(5):785–787
Ben-Hammouda M, Kremer RJ, Minor HC, Sarwar MA (1995) Chemical basis for differential allelopathic potential of sorghum hybrids on wheat. J Chem Ecol 21(6):775–786
Bi F, Huang C, Tong J, Qiu G, Huang B, Wu Q, Li F, Xu Z, Bowser R, Xia XG, Zhou H (2013) Reactive astrocytes secrete lcn2 to promote neuron death. Proc Natl Acad Sci 110(10):4069–4074
Chu YH, Chang CL, Hsu HF (2000) Flavonoid content of several vegetables and their antioxidant activity. J Sci Food Agric 80(5):561–566
Cruz DN, Gaiao S, Maisel A, Ronco C, Devarajan P (2012) Neutrophil gelatinase-associated lipocalin as a biomarker of cardiovascular disease: a systematic review. Clin Chem Lab Med (CCLM) 50(9):1533–1545
Ejoh RA, Djomdi I, Ndjouenkeu R (2006) Characteristics of tigernut (Cyperus rotundus) tubers and their performance in the production of a milky drink. J Food Process Preserv 30(2):145–163
Ellman GL (1959) Tissue sulfhydryl groups. Arch Biochem Biophys 82(1):70–77
Ellman GL, Diane KC, Andres V, Featherstone RM (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7(2):88–95
Fridovich I (1995) Superoxide radical and superoxide dismutases. Annu Rev Biochem 64(1):97–112
Gasmi S, Rouabhi R, Kebieche M, Boussekine S, Salmi A, Toualbia N, Taib C, Bouteraa Z, Chenikher H, Henine S, Djabri B (2017) Effects of Deltamethrin on striatum and hippocampus mitochondrial integrity and the protective role of Quercetin in rats. Environ Sci Pollut Res Int 24:16440–16457
Goetz DH, Holmes MA, Borregaard N, Bluhm ME, Raymond KN, Strong RK (2002) The neutrophil lipocalin NGAL is a bacteriostatic agent that interferes with siderophore-mediated iron acquisition. Mol Cell 10(5):1033–1043
Güven A, Kaya N (2005) Determination of reduced glutathion, glutathione-S-transferase and selenium levels in goose liver cells with damage induced by carbon tetrachloride and ethanol. Turk J Vet Anim Sci 29(6):1233–1238
Haagen L, Brock A (1992) A new automated method for phenotyping arylesterase (EC 3.1. 1.2) based upon inhibition of enzymatic hydrolysis of 4-nitrophenyl acetate by phenyl acetate. Clin Chem Lab Med 30(7):391–396
Halliwell B (1991) Reactive oxygen species in living systems: source, biochemistry, and role in human disease. Am J Med 91(3):S14–S22
Hamed A, Soltan M, Fry J, Hammouda F, Zaki A (2012) Antioxidant and cytoprotective properties of three Egyptian Cyperus species using cell-free and cell-based assays. Pharm Crops 3(1):88–93
Hocine L, Merzouk H, Merzouk SA, Ghorzi H, Youbi M, Narce M (2016) The effects of exposure on biochemical and redox parameters in pregnant rats and their newborns. Pestic Biochem Physiol 134:49–54
Hsu CL, Chen W, Weng YM, Tseng CY (2003) Chemical composition, physical properties, and antioxidant activities of yam flours as affected by different drying methods. Food Chem 83(1):85–92
Hussein J, Mostafa E, El-Waseef M, El-Khayat Z, Badawy E, Medhat D (2011) Effect of omega-3 fatty acids on erythrocyte membrane in diabetic rats. Macedonian J Med Sci 4(3):234–239
Hussein J, Abo Elmatty D, Medhat D, Mesbah N, Farrag AR, Fahmy H (2016) Flaxseed oil attenuates experimental liver hepatitis. Pharm Lett 8(8):142–150
Hussein J, El-Bana M, Refaat E, El-Naggar M (2017) Synthesis of carvacrol-based nanoemulsion for treating neurodegenerative disorders in experimental diabetes. J Funct Foods 37:441–448
Idris SB, Ambali SF, Ayo JO (2012) Cytotoxicity of chlopyrifos and cypermethrin: the ameliorative effects of antioxidants. Afr J Biotechnol 11(99):16461–16467
Imam H, Sofi G, Seikh A, Lone A (2014) The incredible benefits of Nagarmotha (Cyperus rotundus ). Int J Nutr Pharmacol Neurol Dis 4(1):23–27
Jin M, Kim JH, Jang E, Lee YM, Han HS, Woo DK, Park DH, Kook H, Suk K (2014) Lipocalin-2 deficiency attenuates neuroinflammation and brain injury after transient middle cerebral artery occlusion in mice. J Cereb Blood Flow Metab 34(8):1306–1314
Kamala A, Middha SK, Gopinath C, Sindhura HS, Karigar CS (2018) In vitro antioxidant potentials of Cyperus esculentus L. Rhizome extracts and their phytochemical analysis. Pharmacogn Mag 14(54):261–267
Khan AM, Raina R, Dubey N, Verma PK (2018) Effect of deltamethrin and fluoride co-exposure on the brain antioxidant status and cholinesterase activity in Wistar rats. Drug Chem Toxicol 41:123–127
Kilani S, Abdelwahed A, Ammar RB, Hayder N, Ghedira K, Chraief I, Hammami M, Chekir-Ghedira L (2005a) Chemical composition, antibacterial and antimutagenic activities of essential oil from (Tunisian) Cyperus rotundus. J Essent Oil Res 17(6):695–700
Kilani S, Ammar RB, Bouhlel I, Abdelwahed A, Hayder N, Mahmoud A, Ghedira K, Chekir-Ghedira L (2005b) Investigation of extracts from (Tunisian) Cyperus rotundus as antimutagens and radical scavengers. Environ Toxicol Pharmacol 20(3):478–484
Kilani-Jaziri S, Bhouri W, Skandrani I, Limem I, Chekir-Ghedira L, Ghedira K (2011) Phytochemical, antimicrobial, antioxidant and antigenotoxic potentials of Cyperus rotundus extracts. S Afr J Bot 77(3):767–776
Kuda T, Tsunekawa M, Goto H, Araki Y (2005) Antioxidant properties of four edible algae harvested in the Noto Peninsula, Japan. J Food Compos Anal 18(7):625–633
Lee S, Park JY, Lee WH, Kim H, Park HC, Mori K, Suk K (2009) Lipocalin-2 is an autocrine mediator of reactive astrocytosis. J Neurosci 29(1):234–249
Leja-Szpak A, Jaworek J, Tomaszewska R, Nawrot K, Bonior J, Kot M, Palonek M, Stachura J, Czupryna A, Konturek SJ, Pawlik WW (2004) Melatonin precursor, L-tryptophan protects the pancreas from development of acute pancreatitis through the central site of action. J Physiol Pharmacol 55(1):239–254
Manna S, Bhattacharyya D, Mandal TK, Dey S (2005) Neuropharmacological effects of alfa-cypermethrin in rats. Indian J Pharm 37(1):18–20
Mau JL, Chao GR, Wu KT (2001) Antioxidant properties of methanolic extracts from several ear mushrooms. J Agric Food Chem 49(11):5461–5467
Moshage H, Kok B, Huizenga JR, Jansen PL (1995) Nitrite and nitrate determinations in plasma: a critical evaluation. Clin Chem 41(6):892–896
Moustafa YM, Medhat D, Zaitone SA, Ei-Khayat Z, Abdel-Salam OM, Abdalla AA (2018) Amyloid beta-peptide (1-42) induced neurotoxicity in experimental rats: effect of donepezil. Biosci Res 15(3):1931–1942
Nagulendran KR, Velavan S, Mahesh R, Begum VH (2007) In vitro antioxidant activity and total polyphenolic content of Cyperus rotundus rhizomes. J Chem 4(3):440–449
Nieradko-Iwanicka B, Borzęcki A (2015) Subacute poisoning of mice with deltamethrin produces memory impairment, reduced locomotor activity, liver damage and changes in blood morphology in the mechanism of oxidative stress. Pharmacol Rep 67:535–541
Ogaly HA, Khalaf AA, Ibrahim MA, Galal MK, Abd-Elsalam RM (2015) Influence of green tea extract on oxidative damage and apoptosis induced by deltamethrin in rat brain. Neurotoxicol Teratol 50:23–31
Oladipipo AE, Saheed S, Abraham BF (2016) Four weeks daily dose oral administration assessment of Cyperus esculentus L. aqueous extract on key metabolic markers of Wistar rats. Pharmacologia 7(2–3):125–133
Otitoju O, Onwurah IN, Otitoju GT, Ugwu CE (2008) Oxidative stress and superoxide dismutase activity in brain of rats fed with diet containing permethrin. Biokemistri 20(2):93–98
Owen RW, Giacosa A, Hull WE, Haubner R, Spiegelhalder B, Bartsch H (2000) The antioxidant/anticancer potential of phenolic compounds isolated from olive oil. Eur J Cancer 36(10):1235–1247
Pascual JA, Peris SJ (1992) Effects of forest spraying with two application rates of cypermethrin on food supply and on breeding success of the blue tit (Parus caeruleus). Environ Toxicol Chem Int J 11(9):1271–1280
Peerzada AM, Ali HH, Naeem M, Latif M, Bukhari AH, Tanveer A (2015) Cyperus rotundus L.: traditional uses, phytochemistry, and pharmacological activities. J Ethnopharmacol 174:540–560
Polshettiwar SA, Ganjiwale RO, Wadher SJ, Yeole PG (2007) Spectrophotometric estimation of total tannins in some ayurvedic eye drops. Indian J Pharm Sci 69(4):574–576
Ray DE (2001) Pyrethroid insecticides: mechanisms of toxicity, systemic poisoning syndromes, paresthesia, and therapy. In: Krieger R (ed) Handbook of pesticide toxicology. Academic Press, pp 1289–1303
Rice-evans CA, Miller NJ, Bolwell PG, Bramley PM, Pridham JB (1995) The relative antioxidant activities of plant-derived polyphenolic flavonoids. Free Radic Res 22(4):375–383
Romero A, Ramos E, Castellano V, Martínez MA, Ares I, Martínez M, Martínez-Larrañaga MR, Anadón A (2012) Cytotoxicity induced by deltamethrin and its metabolites in SH-SY5Y cells can be differentially prevented by selected antioxidants. Toxicol in Vitro 26:823–830
Saarbrücken SN (2010) Herbal medicine-science embraces tradition: a new insight into ancient Ayurveda. LAP Lambert Academic Publishing, pp 139–148
Sabiu S, Ajani EO, Temitope BO, Bashirat YO (2016) Aqueous extract of Cyperus esculentus L. restores and boosts sexual competence in paroxetine-dysfunctioned male Wistar rats. J Exp Integr Med 6(1):12–20
Sabiu S, Ajani EO, Sunmonu TO, Ashafa AO (2017) Kinetics of modulatory role of Cyperus esculentus L. on the specific activity of key carbohydrate metabolizing enzymes. Afr J Tradit Complement Altern Med 14(4):46–53
Saeed S, Ahmad M, Ahmad M, Kwon YJ (2007) Insecticidal control of the maelybug Phenacoccus gossypiphilous (Hemiptera: Pseudococcidae), a new pest of cotton in Pakistan. Entomol Res 37(2):76–80
Samariya K, Sarin R (2013) Isolation and identification of flavonoids from Cyperus rotundus Linn. In-vivo and in-vitro. J Drug Deliv Ther 3(2):109–113
Sayed HM, Mohamed MH, Farag SF, Mohamed GA (2001) Phytochemical and biological studies of Cyperus rotundus L. growing in Egypt. Bull Pharm Sci Cairo Univ 39:195–203
Seo WG, Pae HO, Oh GS, Chai KY, Kwon TO, Yun YG, Kim NY, Chung HT (2001) Inhibitory effects of methanol extract of Cyperus rotundus rhizomes on nitric oxide and superoxide productions by murine macrophage cell line, RAW 264.7 cells. J Ethnopharmacol 76(1):59–64
Seriolo B, Paolino S, Sulli A, Ferretti V (2006) Cutolo M. Bone metabolism changes during anti-TNF-α therapy in patients with active rheumatoid arthritis. Ann N Y Acad Sci 1069(1):420–427
Shamkuwar PB, Hoshamani AH, Gonjari ID (2012) Antipasmodic effect of Cyperus esculentus L. (Cyperaceae) in diarrhea. Scholars Res Library 4:522–524
Sharma P, Firdous S, Singh R (2014) Neurotoxic effect of cypermethrin and protective role of resveratrol in Wistar rats. Int J Nutr Pharmacol Neurol Dis 4(2):104
Shukla Y, Yadav A, Arora A (2002) Carcinogenic and cocarcinogenic potential of cypermethrin on mouse skin. Cancer Lett 182(1):33–41
Singh KA, Nath Tiwari M, Prakash O, Pratap Singh M (2012a) A current review of cypermethrin-induced neurotoxicity and nigrostriatal dopaminergic neurodegeneration. Curr Neuropharmacol 10(1):64–71
Singh N, Pandey BR, Verma P, Bhalla M (2012b) Gilca M. Phyto-pharmacotherapeutics of Cyperus rotundus Linn. (Motha): an overview. Indian J Nat Prod Resour 3(4):467–476
Singh A, Tripathi P, Prakash O, Singh MP (2016) Ibuprofen abates cypermethrin-induced expression of pro-inflammatory mediators and mitogen-activated protein kinases and averts the nigrostriatal dopaminergic neurodegeneration. Mol Neurobiol 53:6849–6858
Singleton VL, Rossi JA (1965) Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am J Enol Vitic 16(3):144–158
Soderlund DM, Clark JM, Sheets LP, Mullin LS, Piccirillo VJ, Sargent D, Stevens JT, Weiner ML (2002) Mechanisms of pyrethroid neurotoxicity: implications for cumulative risk assessment. Toxicology 171(1):3–59
Thundyil J, Pavlovski D, Sobey CG, Arumugam TV (2012) Adiponectin receptor signalling in the brain. Br J Pharmacol 165(2):313–327
Verschoyle RD, Aldridge WN (1980) Structure-activity relationships of some pyrethroids in rats. Arch Toxicol 45(4):325–329
Wang XP, Xue FS, Hua AI, Ge F (2006) Effects of diapause duration on future reproduction in the cabbage beetle, Colaphellus bowringi: positive or negative? Physiol Entomol 31(2):190–196
Wang XH, Souders CL II, Xavier P, Li XY, Yan B, Martyniuk CJ (2020) The pyrethroid esfenvalerate induces hypoactivity and decreases dopamine transporter expression in embryonic/larval zebrafish (Danio rerio). Chemosphere 243:125416
Watanabe S, Okura T, Kurata M, Irita J, Manabe S, Miyoshi K et al (2006) The effect of losartan and amlodipine on serum adiponectin in Japanese adults with essential hypertension. Clin Ther 28: 1677–1685
Wennberg A, Gustafson D, Hagen CE, Roberts RO, Knopman D, Jack C Jr, Petersen RC, Mielke MM (2016) Serum adiponectin levels, neuroimaging, and cognition in the mayo clinic study of aging. J Alzheimers Dis 53(2):573–581
Zhang HY, Hanna MA, Ali Y, Nan L (1996) Yellow nut-sedge ( Cyperus esculentus L.) tuber oil as a fuel. Ind Crop Prod 5(3):177–181
Zhishen J, Mengcheng T, Jianming W (1999) The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chem 64(4):555–559
Zommara M, Imaizumi K (2017) In Vitro antioxidant activity of chufa tubers (Cyperus esculentus L.) extracts in liposome peroxidation systems. J Sustain Agr Sci 43(2):69–76
Acknowledgments
The authors acknowledge the National Research Centre (NRC), Giza, Egypt, for providing necessary facilities for the accomplishment of this research work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical clearance
Prior ethical clearance for experimentation was approved by the National Research Centre (NRC), Giza, Egypt. In all the experimentation, the ethical care and treatment of the animals were conducted as per the guidelines laid down and following the regulations of the ethical committee of NRC.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hussein, J.S., Medhat, D., Abdel-Latif, Y. et al. Amelioration of neurotoxicity induced by esfenvalerate: impact of Cyperus rotundus L. tuber extract. Comp Clin Pathol 30, 1–10 (2021). https://doi.org/10.1007/s00580-020-03182-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00580-020-03182-0